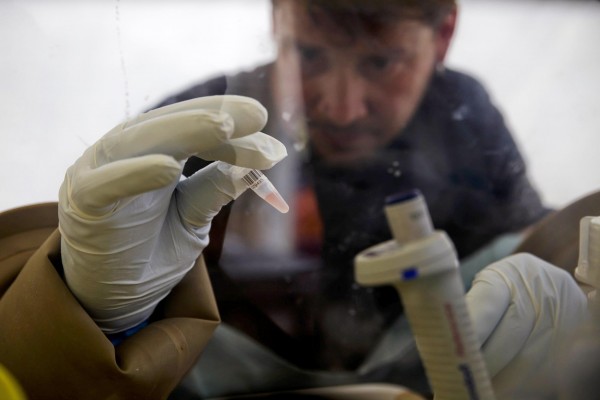
An antibody created by Canadian organization Immunovaccine Inc., was discovered to be defensive against Ebola infection in creatures. Four monkeys contaminated with the Ebola infection made due in the wake of being infused the immunization. The organization announced the study comes about on Monday which came about its stock exchanges to take off high.
The Halifax, Nova Scotia-based organization said that the four monkeys were initially inoculated and later a deadly measurement of Ebola infection was infused in them. While these four creatures survived the viral assault, two monkeys in the control bunch, who did not got the antibody, passed on inside a week.
The organization began the creation of Ebola antibody few months once more after the U.s. National Institutes of Health (NIH) proposed Immunovaccine to create antibody for Ebola actualizing the engineering utilized by the organization within the improvement of Bacillus anthracis immunization.
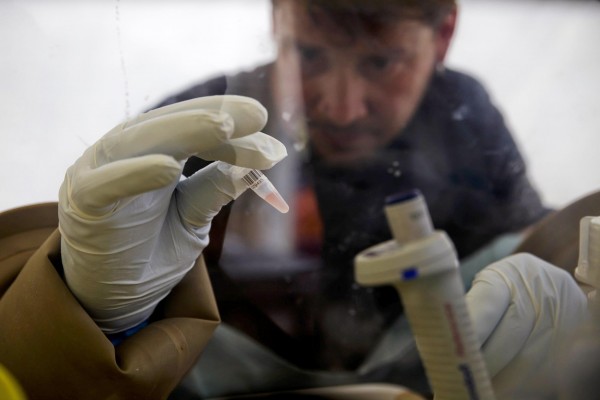
The antibody generation work was a piece of NIH tests on potential antigens, executors (substances) that trigger the invulnerable framework to deliver antibodies against the Ebola infection. The antigen introduce in this immunization does not utilize a same infection to assault the cells dissimilar to antigens of other early-arrange Ebola antibodies.
Presently Zmapp, an exploratory medication is been utilized to treat Ebola infection despite the fact that FDA (Food and Drug Administration) has not affirmed it authoritatively because of absence of human clinical trials.
What is Zmapp and how it functions?
Zmapp, blend medication contains monoclonal antibodies produced using a tobacco-plant strain. It is a streamlined mixed drink consolidating the best segments of two medicines to be specific MB-003 (Mapp) and Zmab (Defyrus/PHAC) and is created in a research facility by laying open mice to pieces of the infection. This medication contains antibodies that give security against the infection assaulting the body. These antibodies tie to a particular protein (focus) of the infection and copy the resistant activity.
Why has the FDA not endorsed Zmapp?
Starting now, just two cases have been accounted for expressing that Zmapp has been viable in treating Ebola, while studies have demonstrated that just 43% of creatures influenced with the Ebola infection have been cured. Also, the adequacy of the medication has not been assessed in people (just two cases have been sure where the medication was utilized before contamination and not in the wake of being influenced).
The medication is constantly utilized focused around the exploratory confirmation and its wellbeing must be demonstrated through clinical trials. It may take no less than two months for huge scale considers on people and in this manner, a lot of time for the FDA to formally favor the utilization of Zmapp for Ebola treatment. Read to know more Facts about Zmapp you ought to know